Abstract
Introduction
Inherited thrombocytopenias (ITs) are a heterogeneous group of rare platelet disorders. which lead not only to increased bleeding, but also to syndromic forms. ITs are caused by genetic alterations in megakaryopoiesis-related genes. In the last years, whole-exome sequencing (WES) has allowed the identification of novel genes involved in IT.
Aim
To perform the molecular, clinical and platelet characterization of two unrelated families with syndromic IT, to unveil the underlaying alteration leading to the disease. To explore the functional role of the identified alterations during megakaryocytic (Mk) differentiation.
Methods
WES was performed in two unrelated non-consanguineous families with lifelong severe macrothrombocytopenia (MCT), bleeding, and extra-hematological manifestations. Bleeding score (BS) was recorded by ISTH-BAT. Platelet phenotyping included platelet count (P), blood film, aggregometry (LTA) and flow cytometry (FC). UDP-galactose-4-epimerase enzymatic activity was measure by HPLC/MS/MS. In vitro functional studies were performed through overexpression of GALE genetic variants in human K562 cell line to elucidate its role in Mk differentiation, by measuring cell ploidy and expression of CD41, CD61 and CD42b surface markers after 7-days of PMA treatment.
Results
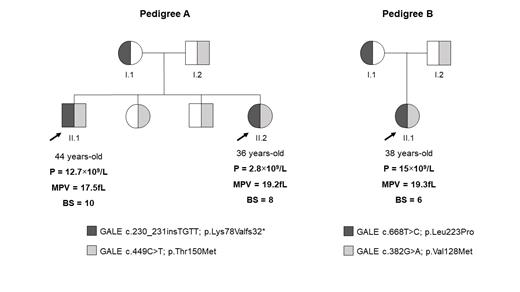
Family pedigrees are shown in Figure 1. Three patients (A.II.1, A.II.2 and B.II.1) were referred due to lifelong severe MCT and moderate-severe bleeding tendency (Figure 1). Moreover, they presented mental retardation, mitral insufficiency, and increased bilirubin levels. Blood film revealed enlarged, giant, and grey platelets (A.II.1: 36%, 6% and 54%, respectively; A.II.2: 56%, 4%, 34%, respectively; B.II.1: 32%, 46%, 12%).
LTA showed moderate/severe impaired aggregation with ADP, TRAP-6, CRP, epinephrine, arachidonic acid, and ristocetin. FC confirmed null secretion of alpha and dense granules in A.II.1, A.II.2 and reduced levels in B.II.1 (7.8%, 8.1%, 28.3% respectively, vs. 51.7% control platelets with ADP 10µM; 10.8%, 7.8%, 36.7% respectively, vs. 95.6% control platelets with TRAP6 25µM).
WES revealed that both pedigrees carried compound heterozygous variants in GALE (NM_001127621.2): c.230_231insTGTT; p.Lys78Valfs*32 (exon 3), and c.449C>T; p.Thr150Met (exon 5) in A.II.1 and A.II.2 patients; and, c.668T>C, p.Leu223Pro (exon 7), and c.382G>A, p.Val128Met (exon 5) in B.II.1 (Figure 1). Enzymatic activity of the GALE-encoded protein UDP-galactose-4-epimerase was severely reduced in the affected patients: both A.II.1, A.II.2 patients had 1.3 μmol/h/g hemoglobin (control: 8.8 μmol/h/g hemoglobin), and B.II.1 patient had 0.6 μmol/h/g hemoglobin (control: 8 μmol/h/g hemoglobin).
Furthermore, in vitro overexpression assays between wild-type GALE and p.Thr150Met, p.Leu223Pro and p.Val128Met variants, confirmed a delayed maturation of Mks upon PMA treatment (at 3, 5 and 7 days). characterized by a significant reduction in the expression of the megakaryocytic surface markers CD41, CD61 and CD42b.
Conclusion
WES has allowed us to identify pathogenic variants in GALE, which were associated with syndromic IT characterized by severe macrothrombocytopenia. Patients harboring these pathogenic variants presented moderate to severe bleeding tendency associated with cardiovascular and neurological abnormalities. Regarding the platelet phenotype, the presence of giant and grey platelets and the absence of both platelet granules were the most remarkable features reported. Moreover, these GALE variants led to an alteration in in vitro Mk maturation, supporting the thrombocytopenic phenotype observed in patients.
Funding
ISCIII (PI17/01966, PI 17/01311, PI20/00926), GRS (GRS2061A/19, GRS2135/A/2020), Fundación Séneca (19873/GERM/15), Fundación Mutua Madrileña (AP172142019), Premio López Borrasca (2019), Grupo Trabajo Patología Hemorrágica-SETH (2020).
Hernández-Rivas: Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene/BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees; Pfizer: Honoraria, Membership on an entity's Board of Directors or advisory committees.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal